 |
 |
| |
Habitats, behaviour, & ecology |
| |
 |
| |
Ghost shrimps
|
| |
Much has been written on habitats, behaviour, and ecology of west-coast mud- and ghost-shrimps, but much less on “true” or caridean shrimps. Considered here are ghost shrimps, while MUD SHRIMPS and "TRUE" SHRIMPS are dealt with in another section.
|
| |
Research study 1 |
| |

  Ghost shrimps Neotrypaea (Callianassa) californiensis inhabit U- or Y-shaped burrows, commonly on sand-flats. One arm of the burrow complex is usually exhalent and is associated with the presence of a sediment mound (see drawing of side view of burrow on Left). Ghost shrimps Neotrypaea (Callianassa) californiensis inhabit U- or Y-shaped burrows, commonly on sand-flats. One arm of the burrow complex is usually exhalent and is associated with the presence of a sediment mound (see drawing of side view of burrow on Left).
Resin casts of burrow structure show several bulbs or chambers used for turning and for storing organic debris such as small pieces of eelgrass Zostera for food (photo on Right).
Burrows in high-density beds show little inter-communication (see drawings lower Left). Drawings from Griffis & Chavez 1988 J Exper Mar Biol Ecol 117: 239; resin-cast photo from Nickell & Atkinson 1995 Mar Ecol Prog Ser 128: 181.
The apparent cavity at the top of the left cluster is
not a common chamber; rather, it appears to be just a
depression in the sand into which the liquid resin was poured |
| |

Research study 2 |
| |
In the Fraser River estuary in British Columbia, densities of Neotrypaea californiensis burrow-openings may reach 350-450 . m-2. The burrows extend 20-50cm into the sediment and then branch horizontally for distances of up to 1m. Each system usually has 2 exits, and contains numerous bulbous turn-arounds and blind alleys throughout. There is no distinct lining to the burrow walls and they are thought to be temporary feeding burrows rather than permanent dwelling burrows. Swinbanks & Murray 1981 Sedimentology 28: 201; Swinbanks & Luternauer 1987 J Paleont 61: 315.
In comparison, burrow densities of ghost shrimps N. californiensis may reach 50-170 . m-2 in Mugu Lagoon, California, 300-700 . m-2 in Oregon, 290-330 in areas of Washington, and 140-180 . m-2 in the Fraser River estuary. Feldman et al. 2000 Estuaries 23: 141.
Several co-inhabiting clams Cryptomya californica are shown,
with their siphons opening into the shrimps' burrows |
| |

Research study 3 |
| |

Numbers of both types of shrimps vary from year to year. The graph shows yearly variation in shrimp densities in Willapa Bay, Washington over a 7yr period. Dumbauld et al. 1996 J Crust Biol 16: 689.
NOTE the data are based on counts of burrow openings, and a conversion factor of 1.5 burrow holes per shrimp for Upogebia and 1.2 burrow holes per shrimp for Neotrypaea |
| |

Research study 4 |
| |
 Reports in the literature differ as to whether mud-shrimp and ghost-shrimp burrows are inhabited by a single individual or a male and female mating pair. Perhaps the 2 sexes only join up during reproductive time, or geographic differences may explain the differences. MacGinitie & MacGinitie 1968 Natural history of marine animals McGraw-Hill, N.Y.; Swinbanks & Murray 1981 Sedimentology 28: 201; Griffis & Chavez 1988 J Exper Mar Biol Ecol 117: 239. Photograph courtesy Dave Cowles, Walla Walla University, Washington www.wallawalla.edu. Reports in the literature differ as to whether mud-shrimp and ghost-shrimp burrows are inhabited by a single individual or a male and female mating pair. Perhaps the 2 sexes only join up during reproductive time, or geographic differences may explain the differences. MacGinitie & MacGinitie 1968 Natural history of marine animals McGraw-Hill, N.Y.; Swinbanks & Murray 1981 Sedimentology 28: 201; Griffis & Chavez 1988 J Exper Mar Biol Ecol 117: 239. Photograph courtesy Dave Cowles, Walla Walla University, Washington www.wallawalla.edu. |
| |

Research study 5 |
| |

In comparison with the more firmly constructed and permanent burrows of mud shrimps, burrows of ghost shrimps Neotrypaea californiensis are unlined and often lack discernible openings to the sand surface during low tide. For these reasons, plus the fact that mud shrimps ventilate their burrows, it is thought that ghost shrimps may be more exposed to hypoxic interstitial waters than mud shrimps. Studies at Hatfield Marine Sciences Center, Oregon show that both species, but especially ghost shrimps, have low metabolic rates, and both tolerate low and even zero (anoxic) oxygen levels for several days if non-moulting. Thompson & Pritchard 1969 Biol Bull 136: 274.
NOTE lit. “low oxygen”; anoxic is “without oxygen”
The degree of "humped-ness" of ghost-shrimp burrows relates to whether the digging is
fresh (top burrow) or older (bottom burrow), and also with the extent of wave action 0.3X |
| |

Research study 6 |
| |
 In a study of space competition in sand-bottom habitats in Mugu Lagoon, California, 6 invertebrate species, including the ghost shrimp Neotrypaea californiensis, are found to account for almost 90% of the macrofaunal suspension-feeders present. The 6 species (4 bivalves, one sand dollar, and one ghost shrimp) are stratified such that their vertical distributions overlap only slightly (see figure on Right). In a study of space competition in sand-bottom habitats in Mugu Lagoon, California, 6 invertebrate species, including the ghost shrimp Neotrypaea californiensis, are found to account for almost 90% of the macrofaunal suspension-feeders present. The 6 species (4 bivalves, one sand dollar, and one ghost shrimp) are stratified such that their vertical distributions overlap only slightly (see figure on Right).

The 3 species that do overlap (the bivalve Nuttallia nuttallii1 and the ghost shrimp/bivalve pair N. californiensi/Cryptomya californica2, treated here as a unit because of their functionally related co-occurrence, are segregated horizontally. The 6 species show relatively little temporal change in their densities over a 3-yr study period. Average density of all macrofauna species over this time is about 600 . m-2, but this rises to about 6000 . m-2 if all benthic fauna is included.
Another part of the study reveals a negative correlation between numbers of Neotrypaea californiensis/Cryptomya californica and the bivalve Nuttallia nuttallii, suggesting competitive exclusion  (see graph lower Right). The generally hyperbolic shape of the graph3 indicates that when either species is common, the other is rare, and only when each is rare are they found in nearly equal frequency. Moreover, when all ghost shrimps and Cryptomya are removed from experimental plots, and then numbers re-assessed later, densities of bivalves Nuttallia and sand dollars Dendraster go up significantly. The author suggests that the ghost shrimps may exclude the clam and sand-dollar species by consuming their larvae and newly settled juveniles, and by burying the juvenile stages. Peterson 1977 Mar Biol 43: 343. (see graph lower Right). The generally hyperbolic shape of the graph3 indicates that when either species is common, the other is rare, and only when each is rare are they found in nearly equal frequency. Moreover, when all ghost shrimps and Cryptomya are removed from experimental plots, and then numbers re-assessed later, densities of bivalves Nuttallia and sand dollars Dendraster go up significantly. The author suggests that the ghost shrimps may exclude the clam and sand-dollar species by consuming their larvae and newly settled juveniles, and by burying the juvenile stages. Peterson 1977 Mar Biol 43: 343.
NOTE1 the small photo in the schematic on the Right is actually of the closely related N. obscurata
NOTE2 the ghost-shrimp burrows extend to 50cm depth and generally have 2 or more openings. By living embedded in the wall of a shrimp’s burrow, Cryptomya effectively increases its depth distribution much more than its short siphons would otherwise allow
NOTE3 all densities are based on areas of 0.06 m2 |
| |

Research study 7 |
| |
 Another way that ghost shrimps might exclude a bivalve species is by clogging their filtering ctenidia with sediments that they stir up when burrowing and deposit-feeding. Thus, one trophic group, the deposit-feeding shrimps, could inhibit the presence of another trophic group, the suspension-feeding bivalves, in a process known as “trophic-group exclusion”. Another way that ghost shrimps might exclude a bivalve species is by clogging their filtering ctenidia with sediments that they stir up when burrowing and deposit-feeding. Thus, one trophic group, the deposit-feeding shrimps, could inhibit the presence of another trophic group, the suspension-feeding bivalves, in a process known as “trophic-group exclusion”.
This concept is tested more generally in Coos Bay, Oregon by comparing abundances of 9 macrofaunal species of deposit-feeding invertebrates over a 2yr period in 3 adjoining areas: 1) within a dense intertidal bed of ghost shrimps Neotrypaea californiensis, 2) within a transitional zone characterised by spotty shrimp occurrence, and 3) outside the bed. The 9 deposit-feeding 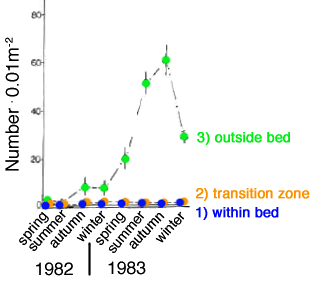 species, representing over 95% of the non-shrimp macrofaunal individuals present, include 3 species of tube-dwelling spionid polychaetes, 3 species of tube-dwelling crustaceans (2 amphipods and one tenaid), and 3 motile species (cumacean, oligochaete, amphipod). species, representing over 95% of the non-shrimp macrofaunal individuals present, include 3 species of tube-dwelling spionid polychaetes, 3 species of tube-dwelling crustaceans (2 amphipods and one tenaid), and 3 motile species (cumacean, oligochaete, amphipod).
The results of seasonal assessments of abundance over a 2-yr period show that 7 of the 9 deposit-feeding species have highest densities when numbers of ghost shrimps are low or intermediate, thus providing evidence of trophic-group exclusion. Results for all species cannot be included, here, but one species, the spionid polychaete Streblospio benedicti, exemplifies the general effects of the shrimips (graph on Right). As expected, because they can run away, the most variable results are shown by the 3 motile species. Otherwise, 5 of the 6 sedentary species solidly support the trophic-group hypothesis. Posey 1986 Mar Ecol Progr Ser 31: 15.
NOTE also known as “trophic-group amensalism”
NOTE densities range from 70-130 individuals . m-2 |
| |

Research study 8 |
| |
 Another type of exclusion mediated by burrowing activities of ghost shrimps Neotrypaea californiensis is that of seagrasses Zostera spp. Researchers from Washington Department of Fish & Wildlife find that in areas of Willapa Bay, Washington treated with the pesticide carbaryl to remove shrimp populations, the seagrass Z. japonica quickly colonises (see photograph). Comparison with untreated areas at the same intertidal level reveals that the exclusion occurs not so much at the time of sprouting of Zostera spores in early spring, but later in the season as the shrimps become more active. Zostera japonica is an introduced species that lives higher in the intertidal region where ghost shrimps are more abundant. The native seagrass Z. marina tends to favour lower-intertidal habitats, but is also responsive, to a lesser extent, to carbaryl spraying. Dumbauld & Wyllie-Echeverria 2003 Aquat Bot 77: 27. Another type of exclusion mediated by burrowing activities of ghost shrimps Neotrypaea californiensis is that of seagrasses Zostera spp. Researchers from Washington Department of Fish & Wildlife find that in areas of Willapa Bay, Washington treated with the pesticide carbaryl to remove shrimp populations, the seagrass Z. japonica quickly colonises (see photograph). Comparison with untreated areas at the same intertidal level reveals that the exclusion occurs not so much at the time of sprouting of Zostera spores in early spring, but later in the season as the shrimps become more active. Zostera japonica is an introduced species that lives higher in the intertidal region where ghost shrimps are more abundant. The native seagrass Z. marina tends to favour lower-intertidal habitats, but is also responsive, to a lesser extent, to carbaryl spraying. Dumbauld & Wyllie-Echeverria 2003 Aquat Bot 77: 27.
One year after treatment of test strips in an area of Willapa Bay, Washington with the pesticide carbaryl, growth of seagrasses Zostera japonica is evident
|
| |

Research study 9 |
| |
 One positive feature of ghost-shrimp burrowing is enhancement of nitrogen fixation, a process identified by researchers at the University of Southern California, Los Angeles for Neotrypaea californiensis at Catalina Island. Nitrification is as important for the health of the soil in a mudflat as it is for soil in a wheatfield. Its deficiency significantly limits productivity. Ghost-shrimps aid in the process by introducing oxygen deep into their burrows by ventilatory activity. The oxygen reacts with ammonium ions produced from the breakdown of atmospheric nitrogen gas (N2) to form nitrate ions (NO3-), which is a form of “bioavailable” nitrogen (see schematic). The paper is rich with sediment chemistry, far too detailed to attempt to include here. Bertics et al. 2010 Mar Ecol Prog Ser 409: 1; see also Bertics et al. 2012 Appl Envir Microbiol 78 (11): 3864. One positive feature of ghost-shrimp burrowing is enhancement of nitrogen fixation, a process identified by researchers at the University of Southern California, Los Angeles for Neotrypaea californiensis at Catalina Island. Nitrification is as important for the health of the soil in a mudflat as it is for soil in a wheatfield. Its deficiency significantly limits productivity. Ghost-shrimps aid in the process by introducing oxygen deep into their burrows by ventilatory activity. The oxygen reacts with ammonium ions produced from the breakdown of atmospheric nitrogen gas (N2) to form nitrate ions (NO3-), which is a form of “bioavailable” nitrogen (see schematic). The paper is rich with sediment chemistry, far too detailed to attempt to include here. Bertics et al. 2010 Mar Ecol Prog Ser 409: 1; see also Bertics et al. 2012 Appl Envir Microbiol 78 (11): 3864.
.
NOTE this is facilitated by nitrogenase enzymes that are present in sulphate-reducing bacteria, common in marine sediments. Nitrogenase catalyses the reduction of molecular nitrogen (N2) to ammonia |
| |

Research study 10 |
| |
To what extent do ghost shrimps Neotrypaea californiensis oxygenate their burrows and surrounding sediments by their pumping activities? This is investigated for shrimps in Yaquina Bay, Oregon using techniques of porewater pressure sensing, time-lapse photography, and oxygen sensing, all experiments being done in laboratory tanks. Results show that shrimps in a head-up orientation are actively pumping about 40% of the time, leading to pressurisation of the burrow, transport of oxygen into the surrounding sediment, and a net flow of anoxic water through the sediment-water interface of the sea bottom . During periods of head-down pumping (1-2 times per hour), however, these flows are reversed, and anoxic sediment water is drawn into the burrow (see schematic below). Such oxic/anoxic oscillations can be detected up to 4cm away from the burrow in more sandy sediments, but only to a few mm in mud sediments. The study contributes greatly to our understanding of the oscillatory bioirrigating effects of burrow-inhabiting macrofauna. Volkenborn et al. 2012 Mar Ecol Prog Ser 455: 141. The authors provide a video of a shrimp constructing its U-shaped burrow filmed at about 5000-fold normal speed http://www.int-res.com/articles/suppl/m455p141_supp/
NOTE the technique used is planar-optode imaging, referenced in the paper |
| |
 |
28h record of porewater pressure in sediment containing a ghost shrimp Neotrypaea californiensis burrow. The positive/ negative frequency is the thing to note, not the magnitude. Magnitude increases with time as the deepening shrimp’s burrow gets closer to the pressure sensor, buried at 15cm depth. An average burrow in the study is 24cm deep, 57cm long, with 7 turning chambers. Mean beat frequency is 22 . min-1 |
| |
|
| |

Research study 11 |
| |
A study by researchers from San Diego State University and University of California, Davis suggest that the interaction between ghost shrimps Neotrypaea californiensis and eelgrass Zostera may not be so one-sided as the account in Research Study 8 above suggests, especially in lower intertidal areas where Z. marina is the potential space-competitor. Through a series of 30wk-long translocation experiments in Tomales Bay the authors find that addition of eelgrass to ghost shrimp-dominated areas causes rapid decline in shrimp densities, and addition of ghost shrimps to  eelgrass-dominated areas results in poor survival or displacement of the shrimps. In the first scenario the eelgrass may do so well that it expands vigorously into surrounding ghost-shrimp areas, leading to further displacement. Additionally, when structural mimics of eelgrass rhizomes and roots are implanted into ghost-shrimp habitats, the shrimps quickly move away, suggesting that the eelgrass may physically constrain the burrowing abilities or needs (e.g., the requirement for turn-around chambers in the burrow) of the shrimps. The researchers conclude that eelgrass habitat is generally resistant to modification by ghost shrimps. If the eelgrass is actually the competitive dominant in the system, how then do the species coexist in some areas? The authors note that when an eelgrass patch is damaged at its edges adult shrimps are able to quickly move into the area. Since such disturbances are common in shallow estuarine habitats, the authors suggest that they may be the primary faciltators of coexistence of the 2 species. Castorani et al. 2014 Ecology 95 (8): 2277. Photograph of eelgrass and ghost-shrimp burrows (below Left) courtesy K.A. Hovel, California. eelgrass-dominated areas results in poor survival or displacement of the shrimps. In the first scenario the eelgrass may do so well that it expands vigorously into surrounding ghost-shrimp areas, leading to further displacement. Additionally, when structural mimics of eelgrass rhizomes and roots are implanted into ghost-shrimp habitats, the shrimps quickly move away, suggesting that the eelgrass may physically constrain the burrowing abilities or needs (e.g., the requirement for turn-around chambers in the burrow) of the shrimps. The researchers conclude that eelgrass habitat is generally resistant to modification by ghost shrimps. If the eelgrass is actually the competitive dominant in the system, how then do the species coexist in some areas? The authors note that when an eelgrass patch is damaged at its edges adult shrimps are able to quickly move into the area. Since such disturbances are common in shallow estuarine habitats, the authors suggest that they may be the primary faciltators of coexistence of the 2 species. Castorani et al. 2014 Ecology 95 (8): 2277. Photograph of eelgrass and ghost-shrimp burrows (below Left) courtesy K.A. Hovel, California.
NOTE other observations are made at Mission Bay near San Diego
NOTE rhizome shapes are first carved into plywood sheets in 2 simulated densities (see photograph below), then cast in polyurethane adhesive |
| |
 |
Boundary between a patch of eelgrass Zostera marina and burrows of ghost shrimps Neotrypaea californiensis |
 |
Plastic rhizome-mimics have nylon-line "roots" glued to
them. The mimics are
buried in test plots
at about 2cm depth |
|
| |

|
| |
RETURN TO TOP |

 Reports in the literature differ as to whether mud-shrimp and ghost-shrimp burrows are inhabited by a single individual or a male and female mating pair. Perhaps the 2 sexes only join up during reproductive time, or geographic differences may explain the differences.
Reports in the literature differ as to whether mud-shrimp and ghost-shrimp burrows are inhabited by a single individual or a male and female mating pair. Perhaps the 2 sexes only join up during reproductive time, or geographic differences may explain the differences. 

 Ghost shrimps Neotrypaea (Callianassa) californiensis inhabit U- or Y-shaped burrows, commonly on sand-flats. One arm of the burrow complex is usually exhalent and is associated with the presence of a sediment mound (see drawing of side view of burrow on Left).
Ghost shrimps Neotrypaea (Callianassa) californiensis inhabit U- or Y-shaped burrows, commonly on sand-flats. One arm of the burrow complex is usually exhalent and is associated with the presence of a sediment mound (see drawing of side view of burrow on Left). 


 In a study of space competition in sand-bottom habitats in Mugu Lagoon, California, 6 invertebrate species, including the ghost shrimp Neotrypaea californiensis, are found to account for almost 90% of the macrofaunal suspension-feeders present. The 6 species (4 bivalves, one sand dollar, and one ghost shrimp) are stratified such that their vertical distributions overlap only slightly (see figure on Right).
In a study of space competition in sand-bottom habitats in Mugu Lagoon, California, 6 invertebrate species, including the ghost shrimp Neotrypaea californiensis, are found to account for almost 90% of the macrofaunal suspension-feeders present. The 6 species (4 bivalves, one sand dollar, and one ghost shrimp) are stratified such that their vertical distributions overlap only slightly (see figure on Right).
 (see graph lower Right). The generally hyperbolic shape of the
(see graph lower Right). The generally hyperbolic shape of the  Another way that ghost shrimps might exclude a bivalve species is by clogging their filtering ctenidia with sediments that they stir up when burrowing and deposit-feeding. Thus, one trophic group, the deposit-feeding shrimps, could inhibit the presence of another trophic group, the suspension-feeding bivalves, in a process known as “
Another way that ghost shrimps might exclude a bivalve species is by clogging their filtering ctenidia with sediments that they stir up when burrowing and deposit-feeding. Thus, one trophic group, the deposit-feeding shrimps, could inhibit the presence of another trophic group, the suspension-feeding bivalves, in a process known as “ species, representing over 95% of the non-shrimp macrofaunal individuals present, include 3 species of tube-dwelling spionid polychaetes, 3 species of tube-dwelling crustaceans (2 amphipods and one tenaid), and 3 motile species (cumacean, oligochaete, amphipod).
species, representing over 95% of the non-shrimp macrofaunal individuals present, include 3 species of tube-dwelling spionid polychaetes, 3 species of tube-dwelling crustaceans (2 amphipods and one tenaid), and 3 motile species (cumacean, oligochaete, amphipod).  Another type of exclusion mediated by burrowing activities of ghost shrimps Neotrypaea californiensis is that of seagrasses Zostera spp. Researchers from Washington Department of Fish & Wildlife find that in areas of Willapa Bay, Washington treated with the pesticide carbaryl to remove shrimp populations, the seagrass Z. japonica quickly colonises (see photograph). Comparison with untreated areas at the same intertidal level reveals that the exclusion occurs not so much at the time of sprouting of Zostera spores in early spring, but later in the season as the shrimps become more active. Zostera japonica is an introduced species that lives higher in the intertidal region where ghost shrimps are more abundant. The native seagrass Z. marina tends to favour lower-intertidal habitats, but is also responsive, to a lesser extent, to carbaryl spraying.
Another type of exclusion mediated by burrowing activities of ghost shrimps Neotrypaea californiensis is that of seagrasses Zostera spp. Researchers from Washington Department of Fish & Wildlife find that in areas of Willapa Bay, Washington treated with the pesticide carbaryl to remove shrimp populations, the seagrass Z. japonica quickly colonises (see photograph). Comparison with untreated areas at the same intertidal level reveals that the exclusion occurs not so much at the time of sprouting of Zostera spores in early spring, but later in the season as the shrimps become more active. Zostera japonica is an introduced species that lives higher in the intertidal region where ghost shrimps are more abundant. The native seagrass Z. marina tends to favour lower-intertidal habitats, but is also responsive, to a lesser extent, to carbaryl spraying.  One positive feature of ghost-shrimp burrowing is enhancement of
One positive feature of ghost-shrimp burrowing is enhancement of 
 eelgrass-dominated areas results in poor survival or displacement of the shrimps. In the first scenario the eelgrass may do so well that it expands vigorously into surrounding ghost-shrimp areas, leading to further displacement. Additionally, when structural
eelgrass-dominated areas results in poor survival or displacement of the shrimps. In the first scenario the eelgrass may do so well that it expands vigorously into surrounding ghost-shrimp areas, leading to further displacement. Additionally, when structural 
